
- Effective Model Risk Management for Stress Testing . The Pre-Congress Masterclass will be led by stress testing and model risk professionals from Darling Consulting Group with further guest practitioners to be.
- After nearly three years of deliberations, regulators finalized the Qualified Residential Mortgage (QRM) rule in October, 2014. The final rule includes a broad definition of QRM and aligns it with the Qualified Mortgage.
ISPE Europe Annual Conference Abstracts. Register Now! Since the publication of the ISPE Drug Shortages Survey report in June 2. This presentation will bring delegates up to date with these.
EMA request for. proposals regarding the prevention of shortages. Speaker: Dr. John Berridge, Strategic Advisor, ISPEFalsified medicines are a growing threat also in Europe. Therefore the European Union has published a. This directive has to be implemented by every member state. In. Germany the secur. Pharm project was founded to design and set up the corresponding national system. The. speaker will introduce the project, talk about the results of the pilot- phase and the lessons learned.
The Analytical Framework. The Analytical Framework is a single platform for data management, valuation, forecasting, hedging, and reporting. All QRM engagements leverage this single, comprehensive application, to deliver true.
Looking for pharmaceutical quality risk management training? Pathwise provides quality risk management for life sciences. Concept to risk management ( In context to Q9) 1. A Brief concept to Risk assessment analysis. We are living in full of risk. Risk management not new.
To. complete the picture, the integration in a European wide network will be shown. Speaker: Dr. Martin Bergen, Managing Director, Secur. Pharm, Germany. Joseph A. De. Feo, 2. 5 years as a Sr.
Consultant, Author and CEO of Juran Global, as well as the successor to. Joseph M. Juran, will provide a view from outside of the pharmaceutical industry. His perspective will. In his engaging style, Mr. De. Feo will discuss and answer these questions: How does Pharma Quality compare with other industries? What capabilities are needed for the leadership role of Quality/Operational Excellence Executives? What are the signs that can show that the industry may not be delivering quality?
How can we integrate the thinking with our metrics discussions? How can Quality by Design work? Speaker: Joe De. Feo, President and CEO, Juran Institute, Inc. If we look back to Biotech in Pharma in a few years, we will recognize that in this year Biopharma entered. On the one hand maturing with much higher productivity, which is urgently.
Pharmacos entering emerging markets with biologics to allow. But also with the need to generate. As in any industry, with maturity comes more. And what has been set up in the past to work globally.
There is a strong need in bioprocessing for a real shift to data driven. We have to make processes much more. And second it is our ethical responsibility to make our.
That is not the case, because we have not been able to develop and manufacture for a cost which would. Innovation globally. There are a lot of real exciting. And the most successful ones. One of the most exciting for me is the s. RNI/m. RNA delivery. I hope we will see continuous positive results in clinical Trialsor cellfree production.
Speaker: Prof. Wolfram Carius, Senior Vice President Biopharma Strategy and Member. Global Leadership Team, Sanofi Frankfurt. An Industry Perspective on the challenges of: Speed to innovation. Health care cost pressures.
Rapidly changing health authority landscape. Increasing competition. Increasing complexity of our supply chains. And how a Quality Culture can become a competitive advantage in the face of these challenges. Speaker: John Pinion, Global Head of PT Quality & Compliance, Roche. In considering what lies ahead for the future of pharmaceuticals, biotechnology and medical devices, this.
The presentation will aim to ask the questions that we need to proactively prepare for on this cultural. Speaker: Carol Bye, Head Pharmaceutical Sciences Quality Assurance Global Quality. Operations, Pfizer.
This presentation will discuss the EMA- FDA pilot program for the Parallel Assessment of Quality by Design. Speaker: Dr. Director, Pfizer Global Quality Strategy. The procedure for implementing Qb. D will be illustrated by a case study where Qb.
D elements such as Real. Time Release Testing were realized for a legacy product. In the presentation the establishment of the design. PAT models will be explained highlighting the encountered.
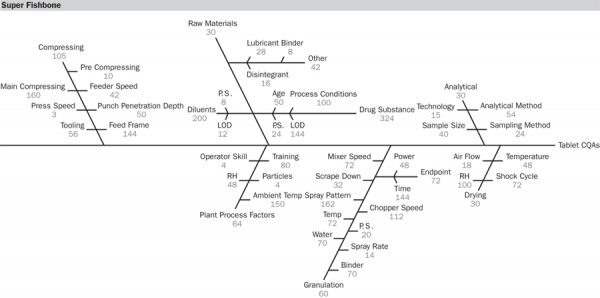
Furthermore lesson learned will be presented as well as the next future steps and projects. Speaker: Dr. Lorenz Liesum, Senior Process Analytical Expert (PAT), Novartis. This presentation addresses the on- going problem of disagreements between industry and regulators in. European procedures. The EU interpretation of the current guidelines will be presented, and the requirements. Finally, some examples of regulatory decisions and the rationale behind them will. Speakers: Dr. Robert Bream, Scientific Administrator, Quality, EMARon Olgivie, Senior Director, Pfizer.
The current global regulatory framework for controlling post- approval CMC changes does not facilitate a. CMC across the product life- cycle, nor does it promote continual.
The life- cycle management of CMC is becoming increasingly important as a result of factors including the. ICH Q8- Q1. 1, globalisation of the development and manufacture of pharmaceuticals, and. A framework for facilitating life- cycle management of CMC which. Increased clarity in what constitutes . David Tainsh, Chief Product Quality Officer, GSKAfter seven years of experience with enhanced (. Everyone sees risks differently.
Based on experience to date, these differences can be observed in the. Lack of clarity and alignment has discouraged industry from introducing Qb. D and enhanced approaches in.
In addition, divergent. US, EU and industry on what details constitute compliance commitments in. CMC lifecycle. management. As a complement to the EMA Qb. D workshop, this presentation introduces constructive opportunities. Speaker: Roger Nosal, VP CGMC, Pfizer Inc. There are differences in the approach to large molecules.
Large molecule processes have always used enhanced approaches to development du to complexity, and. Speaker: Wendy Zwolenski- Lambert, Global Validation Leader, Technical Operations. Novartis. Since more than ten years a discussion is ongoing on the new paradigm in pharmaceutical quality. The. output of this discussion was the adoption of the ICH guidelines on pharmaceutical development (Q8), Quality. Risk Management (Q9), Pharmaceutical Quality System (Q1. Development and Manufacturing of APIs (Q1.
And will. give introduce the recent EMA EFPIA Qb. D workshop which will presented later by both regulator and. Speaker: Dr. Jean- Louis Robert, LNS, France. Jan 2. 01. 4, at EMA with 4. EU agency connected via Web.
Ex, was set up a Qb. D workshop where.
Qb. D application was shared publicly. Each presentation was an opportunity to. Qb. D submission in order to facilitate future.
As one of the key regulatory sponsors of the workshop, Evdokia Korakianiti will share the key. EMA regulator point of view. Speaker: Dr. Evdokia Korakianiti, Head of Procedure Management Department, EMAFor 2. WHO identified convergence as one the key objectives. Very recently, an International Coalition. Medicine Regulatory Agency, including FDA, EMA, China was initiated in order to address the challenge. As a conceptual approach Qb.
D is a cultural change which. Regulatory environment. The presentation will put in.
Speaker: Dr. Georges France, Region Head Quality Europe, Novartis. The European Pharmacopoeia (Ph. Eur.) defines legally binding standards for the quality control of. These standards need to be frequently reviewed to ensure they.
This is why the Ph. Eur. While defining legally- binding standards that will be used as a. Ph. Eur. In doing so, it is. The presentation will provide an overview of measures taken in this direction, including the work of the. Ph. Eur. PAT Working Party.
Speaker: Dr. Susanne Keitel, Director, EDQMThe manufacturing of pharmaceuticals heavily relies on the analytical results generated as in- process. A high number of analytical controls ensures the stability of the.
These analytical. For frequent use in the QC.
The presentation will give an oversight on Analytical Quality by Design (a. Qb. D) Concepts and good method. Opportunities arsing from some elements of the a. Qb. D concepts. within the currently available regulatory framework will be presented.
In particular, the use of post. Finally, consequences for. Speaker: Dr. Oliver Grosche, Governance & Regulations Lead, Novartis. Learn how to change the current paradigm used in the management of quality systems and achieve a 5.
Speakers: Xavier Duburcq, Group BD Director, Altran. Olivier Depardieu, Consulting Partner, Oxo Pharma. ICH Q9 (Quality Risk Management) has been around years.
It highlighted the importance of intelligent. Eight years on how has the pharma industry.
The answer for many is . How can this be? Many companies focus on reactive risk assessment, not proactive risk management to improve operational.
Others have used risk assessment as a . This interactive session will describe the Top Ten . Drawn from over 1,0. Quality Risk Management for business benefit. Speaker: Martin Lush, Vice President, NSF- DBA Ltd. QRM, a regulatory requirement, successfully integrated into the business, drives scientific rigour.
Specifically the presentation will consider the integration of QRM into the European guidance. John Kerridge, Quality Leader, EU Quality Assurance Systems and External. Advocacy, Eli Lilly.
Updates from current PQLI PV Implementation Team activities including: Use of statistics in the validation lifecycle. Validation of biotech processes.
Thoughts on Annex 1. Speaker: Wendy Zwolenski- Lambert, Global Validation Leader, Technical Operations. Novartis. Quality Risk Management (QRM) principles described in ICH Q9 are fully applicable to the control of cross. The revised. versions of chapters 3 and 5 of the EU GMP Guideline state that dedicated facilities are required for.
Such threshold. values have to be derived from toxicological evaluations of the products being manufactured. For this. purpose, the CHMP/Safety Working Party (SWP) developed a draft guideline on setting health based exposure. The. presentation shows how QRM principles can be applied to controlling cross contamination, explains basic.
Speaker: Dr. Hoffmann- La Roche Ltd. What is the true cost of compliance for both industry and the public?
Through using some real life case. Strategies to help companies seek a better path of compliance and also recent. Speaker: David Churchward, Expert GMDP Inspector, MHRAHow does a national regulator use Quality Risk Management approaches? This presentation will explore how.






















 RSS Feed
RSS Feed
